
Description of the Experiment: We have recently developed a new laboratory experiment dealing with the formation and study of metal-organic network compounds. This new experiment is colorful, inexpensive, and illustrates many important chemical principles. The networks are prepared from CuCl, CuBr, and CuI (CuX) with 4,4'-bipyridyl (Bpy). Two different two-dimensional network are formed: (CuX)(Bpy) and (CuX)2(Bpy). Portions of the network structures are shown below:
(CuX)(Bpy)

(CuX)2(Bpy)
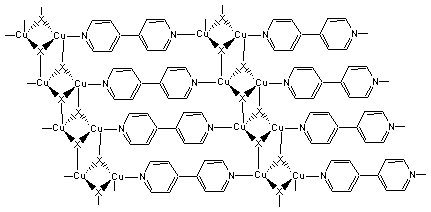
There are three parts to the exercise. Part A involves the synthesis of (CuX)(Bpy) and (CuX)2(Bpy). Part B is a study of the equilibrium between the two products. Part C is an analysis of the compounds from Part A for their copper content by spectroscopy. The details of the new experiment will be published in the Journal of Chemical Education (October, 2001). Some pictures are shown below:



On the left is a solution of CuBr in acetonitrile and solid Bpy in the small beaker. In the center photo, the Bpy has been added to the copper solution and swirled creating the 1:1 (CuCl)(Bpy) product as a red precipitate. In the right hand photo are all six products after precipitation. Mixtures with threefold excess of CuX produce the yellow 2:1 (CuX)2(Bpy) product and those with a deficiency of CuX yield the red 1:1 (CuX)(Bpy) product.


Shown above is the filtration step and the six products as solids (2:1 at the top, 1:1 at bottom, CuCl, CuBr, and CuI, left to right).



Shown on the left is the preparation of the ten mixtures. Note that all of the precipitates are yellow, indicating the formation of 2:1 product in all cases. This is a kinetic product. The middle and left pictures are taken after stirring the mixtures for 5 and 30 minutes, respectively. The conversion of the more Bpy-rich mixtures to the red 1:1 product is evident.

Shown above are the mixtures at the one hour completion point. Those
on the left, which have an excess of CuI, show the unconverted 2:1 product.
The samples on the left, which have an excess of Bpy, have converted to the
red 1:1 compound. The orange samples in the middle contain mixtures of the
two products.



The photo on the left shows a sample of one of the products from Part A being reacted with nitric acid. In the center picture, the mixture is being diluted with ammonia buffer. After dilution to volume, the solution shown next to the Spectronic-20 in the right hand photo is produced. The standards in this picture are made by dilutions of a standard solution of copper(II) to volume with buffer. The absorbance values of the Cu(NH3) 42+ solutions are measured at 610 nm and yield a linear plot. The data allows determination of mass percent copper in the Part A samples.
If you are a college or university instructor, I hope that you will consider
instituting this experiment in your curriculum. Please write to me if you
have questions or comments.